The Science Behind Graphene Oxide
Scientists in the aftermarket auto parts world are constantly looking for newer, better ways to create products that will astonish end users just like you. It doesn’t matter if it’s a car shampoo, wax, detailer spray, or ceramic coating. These chemists have a world of materials at their fingertips, and we (the end-users) are the prime beneficiary of their extraordinary discoveries.
Graphene oxide is the latest chemical compound structure that is quickly taking the market by storm for a couple of reasons. For starters, graphene oxide is highly efficient at shedding water, offering a wide range of practical applications for auto detailing. Additionally, the chemical compound is becoming more readily available via multiple chemical manufacturers.
Everything we know about graphene oxide feels new, interesting, and presents a world of possibilities. That’s why we will detail the fundamental science behind graphene oxide in an easily digestible format here in this blog. Come with us as we uncover a basic introduction to the chemical compound so you can get a better understanding of its most advantageous applications.
Before we can fully dive into graphene oxide, it’s important first to note the differences between graphite and graphene. The former comprises several layers of connecting carbon atoms in a hexagonal shape that resembles chicken wire. Out of all of these countless layers, graphene accounts for only one of them. Think of graphene as an individual playing card where graphite serves as the entire deck.
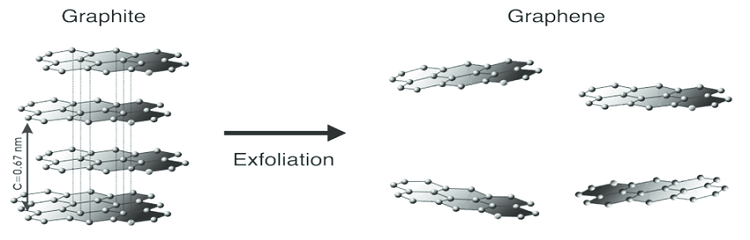
Image Source: Researchgate.net
What’s even more incredible is the fact that graphene is only one atom thick yet still yields a high level of resistance, among other practical properties. Additionally, graphene is flexible, transparent, and it’s so impenetrable that even the smallest atoms will not be able to penetrate its surface.
The process of functionalizing graphene with different chemical groups is where the fun really begins. In fact, functionalizing graphene with oxygen and helium is what creates graphene oxide (GO), the magic material that everyone seems to be talking about in the aftermarket automotive space.
Graphene oxide is slightly more polar than graphene but is still technically non-polar due to its cyclohexane rings. This is best explained through an analogy of a Chinese finger trap. The arrangement of the carbon atoms gives the molecule its strength, which is nearly impenetrable because there are strong reinforcements spread through the entire structure. The hydrogen bonding in the GO allows it to interlink during curing, and the non-polar carbon rings repel water. This chemical reaction is the cornerstone of why water beads off surfaces that are treated with GO.
Water shedding is caused by an incompatibility between water and the coating’s surface. Therefore, if you apply an auto detailing product that contains GO to the surface of your car, it creates a desired water-beading effect. For example, when rain comes into contact with treated areas, you’ll notice that it doesn’t want to stick to the surface.
Also, functionalizing graphene into graphene oxide allows it to have dipole interactions, but non-polar behavior is still more prominent. Dipole interactions occur when the partial charges formed within one molecule are attracted to an opposite partial charge in a nearby molecule, according to Lumen Boundless Chemistry. It’s important to note that the non-polar behavior keeps the electrical charges evenly distributed across the molecule.
Another benefit that chemists seem to enjoy about graphene oxide is the fact that it can be reduced. So, if you have graphene oxide, you can remove the oxygen component once again to create reduced graphene oxide (r-GO). However, r-GO is a different compound with subsequent properties that do not produce the desired effect you would want in an aftermarket automotive product. The chemical compound has weaker binding agents, which is why it’s best to use graphene oxide, aka “the real deal.”
To sum up what we’ve learned today, graphene oxide is a versatile chemical compound with several practical applications. It is an incredibly strong and elastic material that offers a wide variety of uses. Graphene oxide also has non-polar characteristics that make it nearly impossible to dissolve into any solution – not just water
When you consider all of these details, it’s no wonder why R&D chemists are leveraging the power of graphene oxide. The practical chemical compound can serve as a key ingredient for products that will benefit the aftermarket auto industry or any other vertical with the ambition to find a constructive way to utilize it.
Sources:
- The Graphene Handbook 2021 Edition by Ron Mertens
- https://www.nixenepublishing.com
- https://linx-global.com/blog/2018/10/1/graphene-the-worlds-super-material
- https://www.graphene-info.com/reduced-graphene-oxide-introduction
- https://www.nature.com/articles/s41467-020-15381-y
- https://www.researchgate.net/publication/351220163_Magnetic_Characteristics_of_Graphene_Oxide_and_Reduced_Graphene_Oxide
- https://courses.lumenlearning.com/boundless-chemistry/chapter/intermolecular-forces






